
RNF122 suppresses antiviral type I interferon production by targeting RIG-I CARDs to mediate RIG-I degradation | PNAS

Foot-and-Mouth Disease Virus 3B Protein Interacts with Pattern Recognition Receptor RIG-I to Block RIG-I–Mediated Immune Signaling and Inhibit Host Antiviral Response | The Journal of Immunology

A RIG-I–like receptor directs antiviral responses to a bunyavirus and is antagonized by virus-induced blockade of TRIM25-mediated ubiquitination - Journal of Biological Chemistry

Stratified ubiquitination of RIG-I creates robust immune response and induces selective gene expression
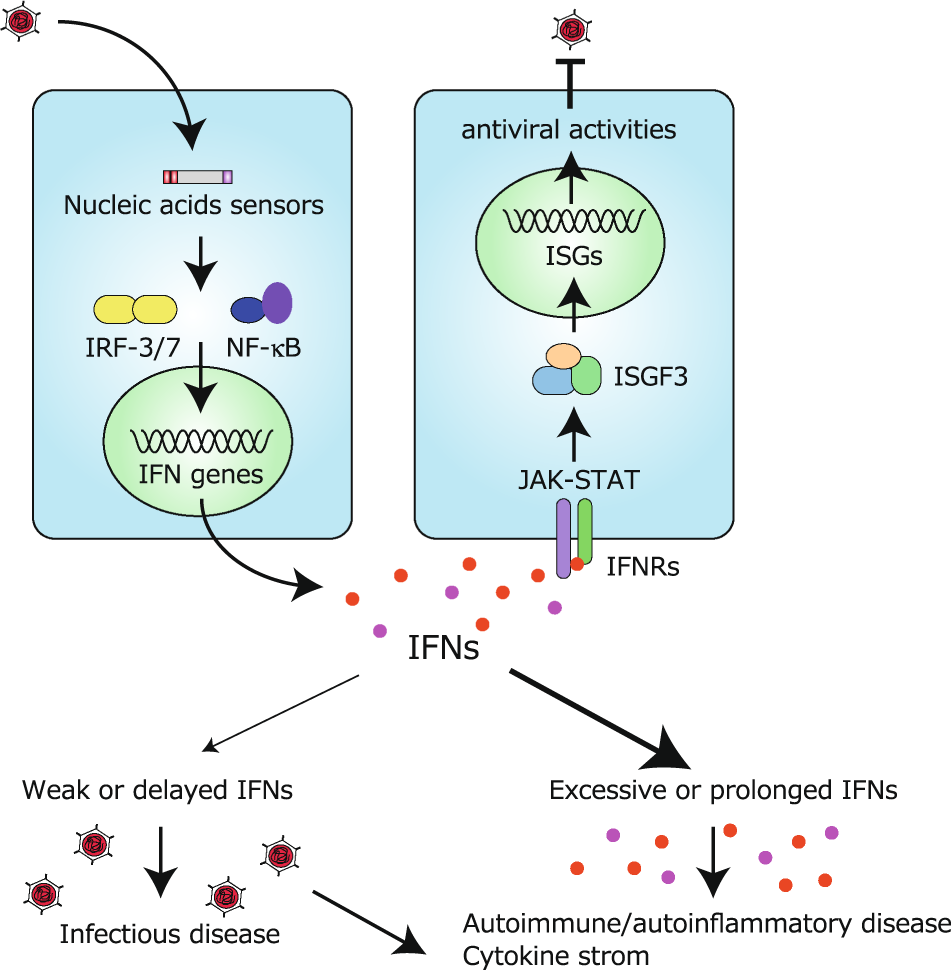
Regulation of RIG-I-like receptor-mediated signaling: interaction between host and viral factors | Cellular & Molecular Immunology

The C-Terminal Domain of Salmonid Alphavirus Nonstructural Protein 2 (nsP2) Is Essential and Sufficient To Block RIG-I Pathway Induction and Interferon-Mediated Antiviral Response | Journal of Virology

SARS-CoV-2 ORF9b inhibits RIG-I-MAVS antiviral signaling by interrupting K63-linked ubiquitination of NEMO - ScienceDirect

RIG-I Forms Signaling-Competent Filaments in an ATP-Dependent, Ubiquitin-Independent Manner - ScienceDirect

Structural insights on dsRNA-mediated activation of RIG-I. RIG-I is an... | Download Scientific Diagram

Riplet/RNF135, a RING Finger Protein, Ubiquitinates RIG-I to Promote Interferon-β Induction during the Early Phase of Viral Infection* - Journal of Biological Chemistry











